BREAST CANCER – AVAILABILITY AND ACCESS
Understanding Breast Cancer Treatment Access Through Named Patient Programs: Availability and Access Pathways
Introduction
Breast cancer is one of the most prevalent cancers affecting women globally, with more than 2.3 million new diagnoses annually. In India, breast cancer represents the second leading cause of cancer deaths in women. For patients diagnosed with advanced or metastatic breast cancer, access to appropriate treatment options can significantly impact survival and quality of life.

What is a Named Patient Program (NPP)?
A Named Patient Program is a regulated pathway that allows patients with serious medical conditions to access medications not yet approved for general use in India. In India, NPP operations are governed by the Central Drugs Standard Control Organization (CDSCO) under the Drugs & Cosmetics Rules, 1945.
This program enables patients with advanced breast cancer to access innovative therapies that have shown promise internationally when standard Indian-approved treatments are not suitable or have been tried without adequate benefit.
How Does NPP Work?
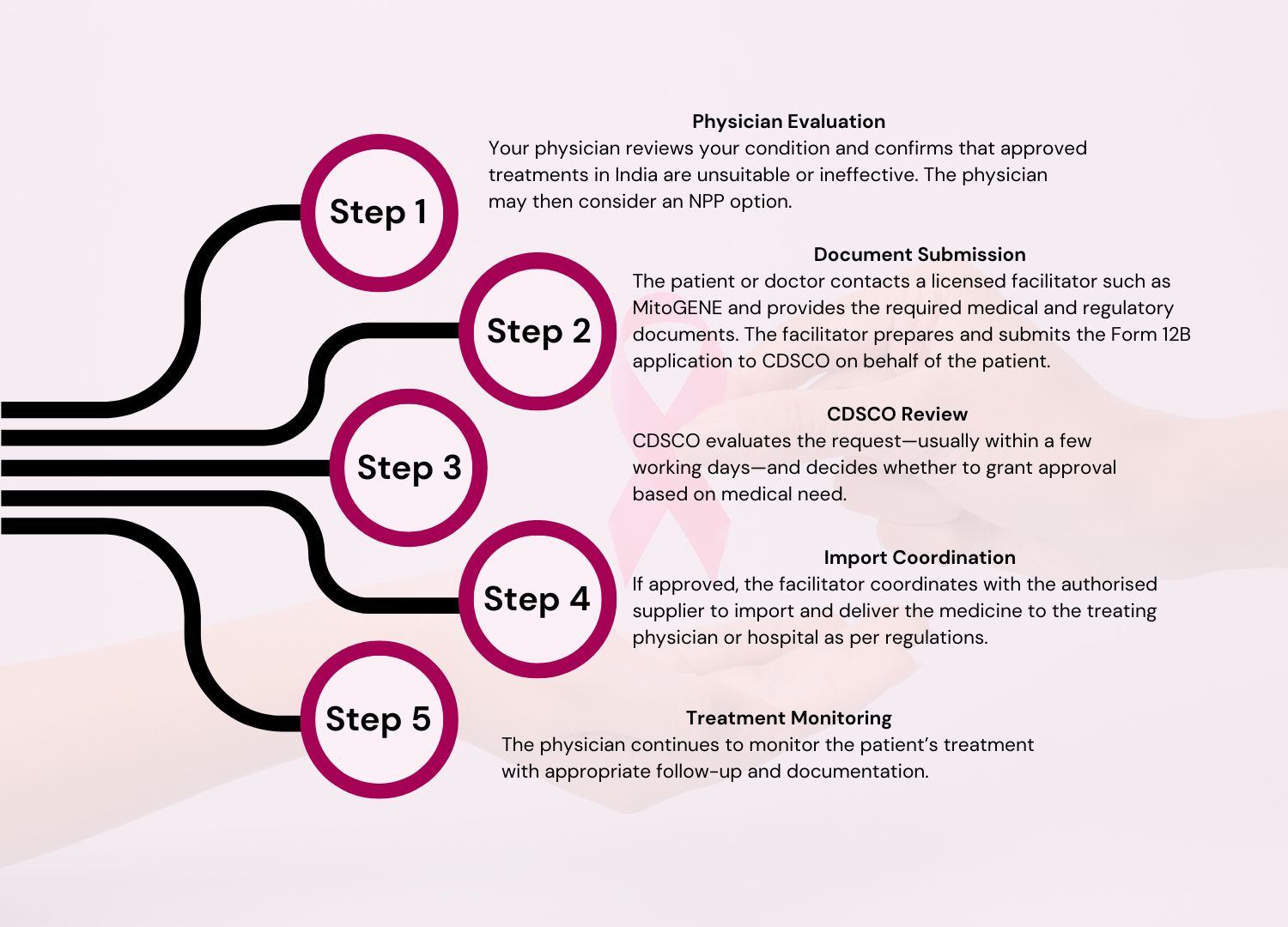
Understanding Breast Cancer Classification
Modern breast cancer diagnosis involves more than confirming the disease is present. Doctors determine:
- Type of cancer: Invasive ductal carcinoma, invasive lobular carcinoma, or other subtypes
- Stage: How far the cancer has spread (localized, regional, or metastatic)
- Molecular characteristics: Testing for hormone receptors (estrogen and progesterone), HER2 status, and other biomarkers
- Overall health status:Ability to tolerate systemic treatment
This comprehensive assessment helps doctors recommend personalized treatment approaches.
Eligibility for NPP Access
To qualify for NPP, breast cancer patients typically must meet these criteria:
- Advanced or metastatic disease requiring urgent systemic intervention
- Exhaustion of suitable Indian-approved alternatives or inadequate response to standard treatments
- Medical evidence supporting potential benefit from the requested medication and it is approved in any other country especially USFDA or EMA
- Clear clinical justification by their treating physician
- Informed consent after understanding potential risks and benefits
Available Breast Cancer Treatment Options
Several systemic therapy approaches are used to treat advanced breast cancer:
Targeted Therapies for HER2-Positive Disease
For patients whose cancer shows high HER2 protein expression, HER2-directed therapies have revolutionized treatment outcomes:
- Tukysa (tucatinib) – for advanced HER2-positive disease
- Nerlynx (neratinib) – as extended therapy for HER2-positive breast cancer
- Lapatinib (available internationally) – a HER2-targeted kinase inhibitor
PARP Inhibitors
These medications are used for patients with specific genetic features, particularly BRCA mutations:
- Talzenna (talazoparib) – for gBRCA-positive, HER2-negative metastatic disease
Hormone Receptor-Positive Disease Treatments
For patients with hormone receptor-positive breast cancer:
- Orserdu (elacestrant) – an estrogen receptor degrader for hormone receptor-positive disease
- Truqap (capivasertib) – a PI3K pathway inhibitor for advanced disease
- Other hormone therapies and CDK4/6 inhibitors available internationally
Antibody-Drug Conjugates (ADCs)
These novel targeted agents combine antibodies with chemotherapy:
- Datroway (datopotamab deruxtecan-dlnk) – for advanced hormone receptor-positive disease
Immunotherapy Options
Checkpoint inhibitors are available for specific breast cancer subtypes, particularly triple-negative breast cancer, enhancing the body’s immune response against cancer cells.
The Role of Advocates Like MitoGENE
Organizations like MitoGENE facilitate the NPP process by:
- Educating patients and physicians about NPP and eligibility
- Facilitating patients to get 12 B import permit for personal use from CDSC
- Connecting patients with legitimate international medicine sources
- Ensuring regulatory compliance
- Providing documentation support for NPP applications
- Coordinating logistics for legal importation and delivery
By working with MitoGENE, patients access expertise and connections necessary to navigate NPP successfully.
“Plz Note: All clinical decisions—including whether a therapy is suitable—are made solely by the treating physician and MitoGENE does not influence prescribing decisions or the medical judgment of physicians”
Conclusion
The Named Patient Program provides an important mechanism for patients with advanced lung cancer to access medications not yet widely available in India. For patients who have exhausted standard Indian treatment options, NPP may provide an opportunity to access promising medications available internationally.
MitoGENE’s expertise as an experienced NPP facilitator significantly simplifies this complex process, providing patients and physicians with knowledgeable guidance and logistical support.
Disclaimer
MitoGENE facilitates patient-specific access to medicines under Named Patient Program permitted by CDSCO through Form 12B. The products mentioned are not approved for commercial sale in India. Access is strictly for personal use under physician supervision. Information provided on this website is sourced from publicly available references, and MitoGENE is not responsible for its completeness or accuracy. MitoGENE does not promote or sell unapproved drugs.
