This medication requires a valid prescription. Please refer to the ‘Prescription Guide’ for details.
Note: We can assist only if the medicine is unavailable in your country.”
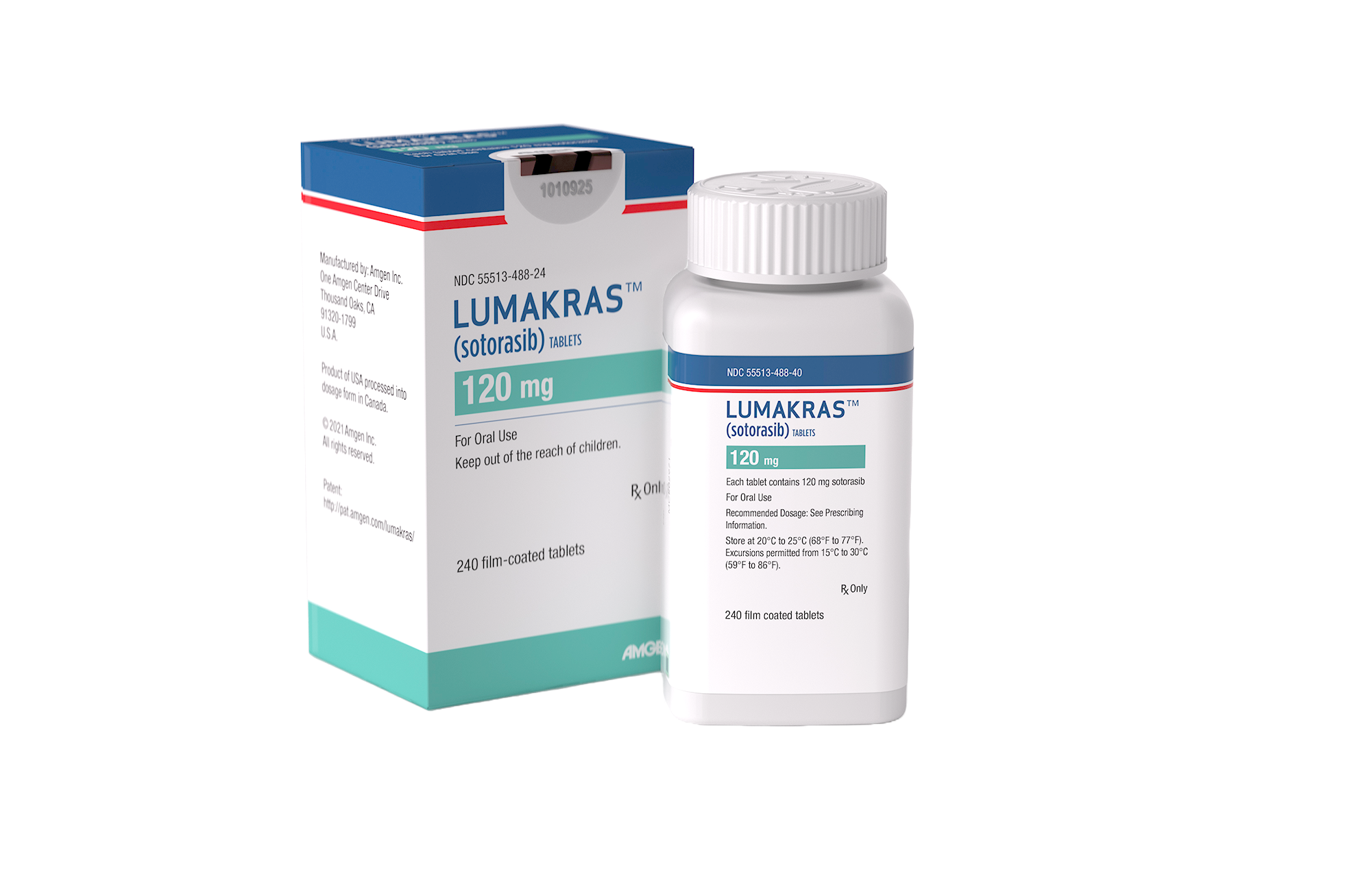
Product Description:
LUMAKRAS (Sotorasib) is a prescription medication indicated for the treatment of adults with KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) who have previously received at least one systemic therapy.
*In the European Union, sotorasib is approved by the EMA under the brand name Lumykras.
Access LUMAKRAS (Sotorasib) in India
LUMAKRAS (Sotorasib) has been approved in countries like the U.S. and Europe but is not yet available for commercial sale in India. However, it is accessible through the Named Patient Program (NPP) for eligible patients.
MitoGENE helps Indian patients access LUMAKRAS (Sotorasib) legally and safely under their doctor’s supervision. We handle the documentation, regulatory approvals, and logistics to ensure compliance with Indian regulations.
If you or your loved one may benefit from LUMAKRAS (Sotorasib), MitoGENE is here to support your journey to access this treatment.
Disease Indications: Lung Cancer
Manufacturer: Amgen GmbH (DE)
Shipping: Room Temperature Shipping. This medication is shipped using standard delivery methods, ensuring it remains within a controlled room temperature range of 15°C to 25°C throughout transit.
Available Dosage Form& Package
- 240 Tablets of 120 mg
Medicine Approved by:
- European Medical Agency (EMA)
- Food and Drug Administration (FDA)
- Medicines & Healthcare products Regulatory Agency (MHRA)




